Background Requirements For Milling API
API Milling is a key step in many drug development processes. Advantages of milling vs. crystallization as the final drug substance unit operation include:
-
Reaching particle size distributions not attainable via crystallization
-
Normalization - ensuring consistent drug substance physical properties and quality for drug product unit operation.
Traditionally, dry milling technologies such as media mills, pin mills, and jet mills have been employed but have come under increased scrutiny due to:
-
Yield losses
-
Industrial hygiene concerns & corresponding containment costs
-
Process train portability
-
Physical property instability occurs due to surface defects, amorphous phase generation, etc.
Wet milling equipment, such as high-pressure homogenizers and media mills, offer a solution to many of the challenges associated with dry milling, albeit they are restricted by low capacities, high energy consumption and frequent maintenance requirements. On the other hand, traditional rotor-stator technology methods can only yield a practical lower mean volume (mV) of ~30 microns (compared to <5 microns via jet milling and ~ 10 microns for dry pin milling).
Quadro's API Wet Milling Solution
Quadro Engineering has developed a new, alternative approach to wet milling with the Quadro® HV-Emulsifier. Via independent testing, it was determined that Quadro’s HV offered more than fifty-five times (55x) the high shear energy of a traditional rotor-stator wet mill.1 Results also demonstrated precise process scaling from lab formulation to pilot and production processing.
During field tests across multiple compounds with a range of starting particle sizes, morphologies and mechanical properties, the following was noted:
-
API size reduction achieved a mean volume (mV) particle size of between 12 - 17 micron for compounds with typical starting PSD’s of between 84 - 179 micron.
-
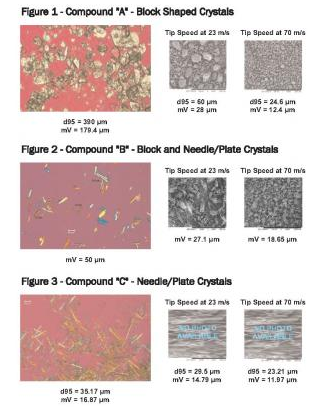
Increased tip speed achieved an additional 30 - 60% reduction of mV & d95 for a wide range of APIs – whether the starting crystals were needle, plate or block-shaped (see figures 1 through 3).
-
Testing did not indicate additional fines generation at elevated tip speeds.
-
Tip velocity appeared to have more size reduction impact on block-shaped compounds than needle/plate crystals, with reduction efficiency averages ranging from 81% - 29%, respectively (reduction efficiency calculated as a percentage of Final PSD/Incoming PSD). Crystals with combined aspect ratio shapes (combination of block and needle/plate crystals) were reduced at an average efficiency of 61%.
-
Lab-scale model (HV0) could be run using significantly small quantities of API materials. This enabled numerous tests to be performed with a lower percentage of APIs (i.e. 20 mg/ml slurries) whilst still achieving a PSD profile that matched full formulation API slurries.
Conclusions
The restricted capacity, high energy consumption, and frequent maintenance requirements of High-Pressure Homogenizers (HPH) and media mills can now be overcome with the Quadro Ytron® HV-Emulsifier thus maximizing productivity while cutting development and production costs.
Related Reading:
- The Benefits of Wet Milling APIs
- Pharmaceutical Mixing Equipment
- Deagglomeration & Wet Milling Equipment
- Milling API Slurries
1An Evaluation of an Enhanced Wet Milling Technology for Further API Particle Size Reduction; AAPS 2011; Ivan Lee(1) Jonathan Ostrosky(2) Mark Walker(3) Eric Sirota(1) Luke Schenck(1) Chemical Process Development and Commercialization(2) Penn State University(3) MMD Finance
High-Shear Rotor–Stator Wet Milling for Drug Substances: Expanding Capability with Improved Scalability | Organic Process Research & Development (acs.org)
Topics:
Emulsification

